Effectiveness of Vaccination and Previous Infection Against Omicron Infection and Severe Outcomes in Children Under 12 Years of Age | medRxiv

Comparative effectiveness of bivalent BA.4-5 and BA.1 mRNA booster vaccines among adults aged ≥50 years in Nordic countries: nationwide cohort study | The BMJ
Vaccines | Free Full-Text | Vaccine Confidence and Uptake of the Omicron Bivalent Booster in Tennessee: Implications for Vulnerable Populations

Comparative effectiveness of bivalent BA.4-5 and BA.1 mRNA booster vaccines among adults aged ≥50 years in Nordic countries: nationwide cohort study | The BMJ
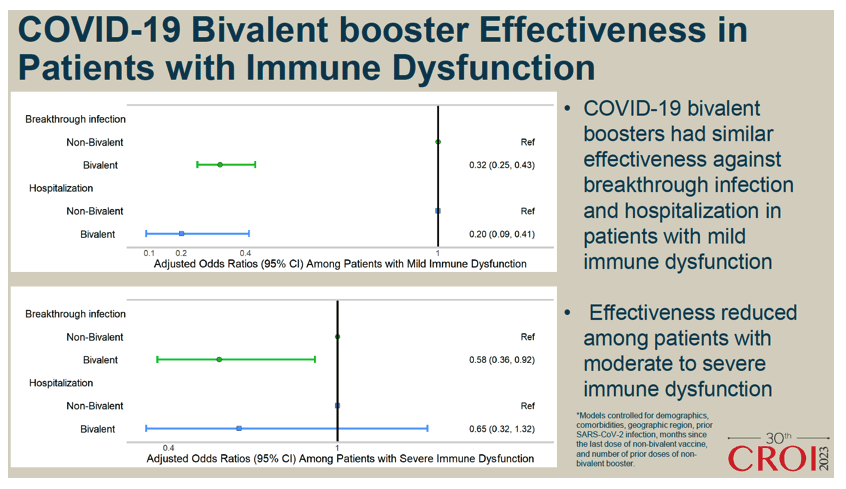
COVID-19 BIVALENT BOOSTER EFFECTIVENESS IN PEOPLE WITH AND WITHOUT IMMUNE DYSFUNCTION - CROI Conference

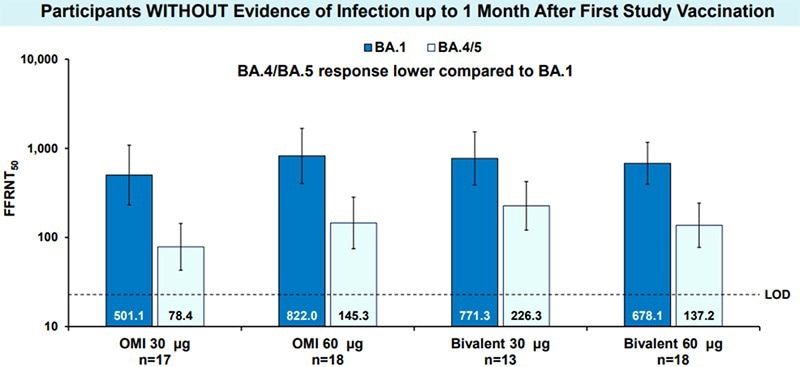
Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...

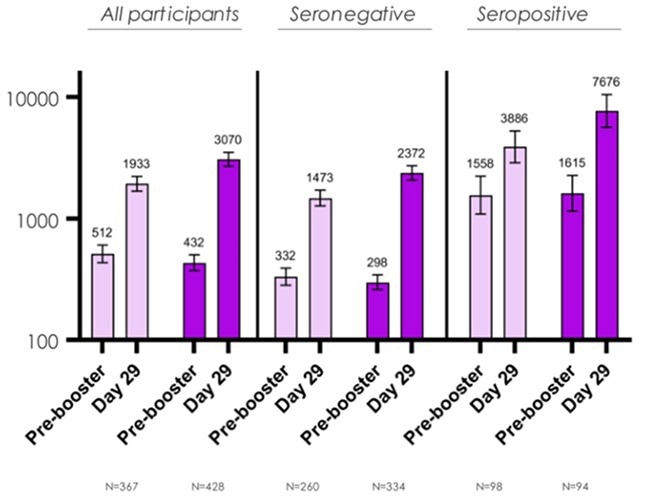
Relative effectiveness of monovalent and bivalent mRNA boosters in preventing severe COVID-19 due to omicron BA.5 infection up to 4 months post-administration in people aged 60 years or older in Italy: a

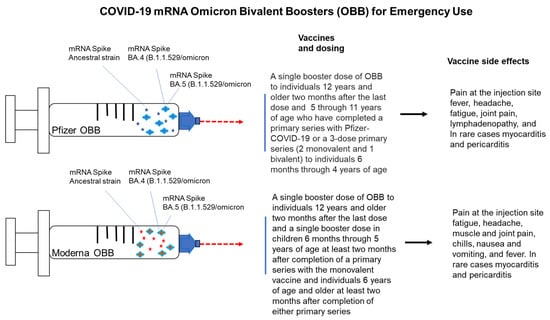
ATAGI recommendations on use of the Pfizer bivalent (Original/Omicron BA.4/5) COVID-19 vaccine | Australian Government Department of Health and Aged Care

Effectiveness of Bivalent mRNA Vaccines in Preventing Symptomatic SARS-CoV-2 Infection — Increasing Community Access to Testing Program, United States, September–November 2022 | MMWR

Updated COVID-19 booster targets subvariants responsible for 98% of cases today : Oregon Health News Blog

Effectiveness of a bivalent mRNA vaccine booster dose to prevent severe COVID-19 outcomes: a retrospective cohort study - The Lancet Infectious Diseases

DPH Shares Monthly COVID-19 Update, Announces Changes to Data Reporting and Booster Guidance - State of Delaware News

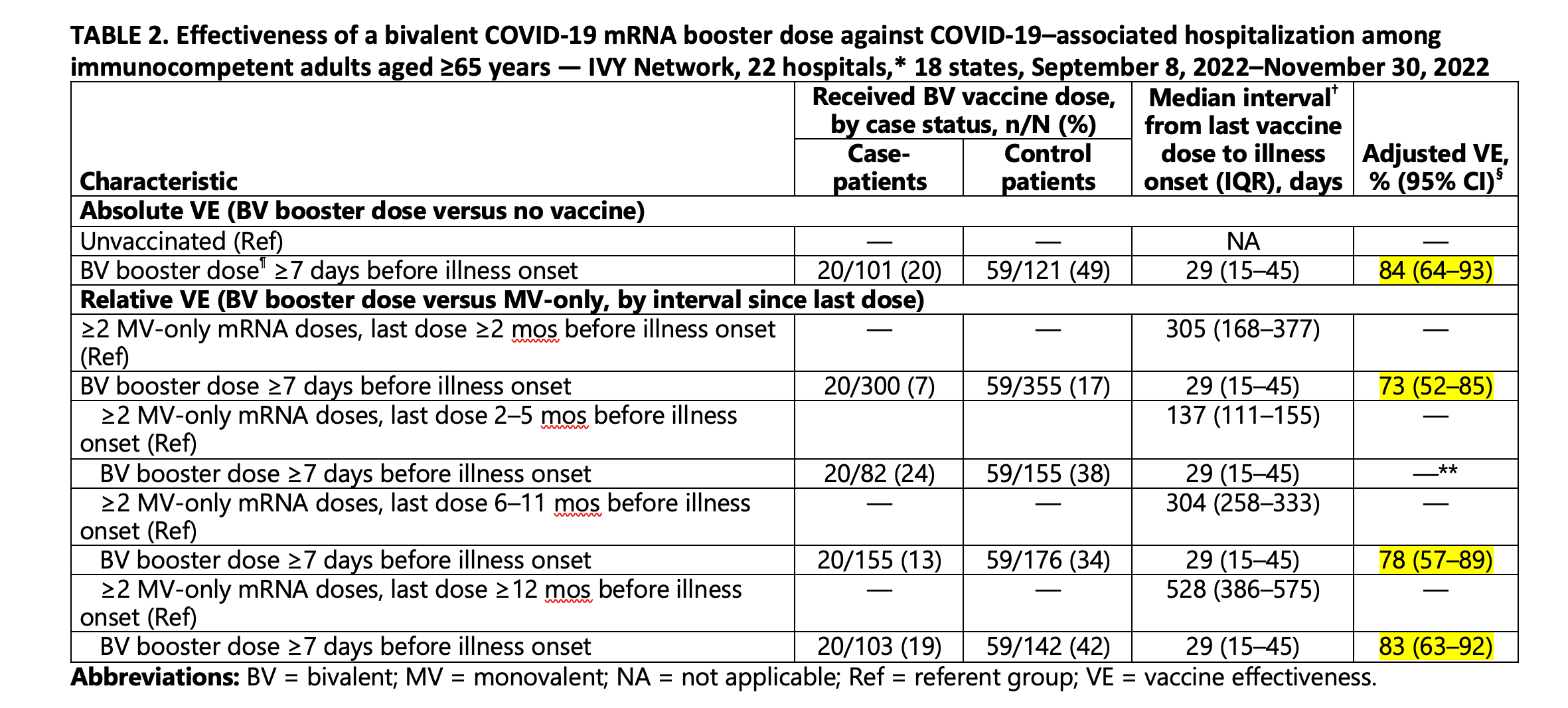
Early Estimates of Bivalent mRNA Vaccine Effectiveness in Preventing COVID-19–Associated Emergency Department or Urgent Care Encounters and Hospitalizations Among Immunocompetent Adults — VISION Network, Nine States, September–November 2022 | MMWR



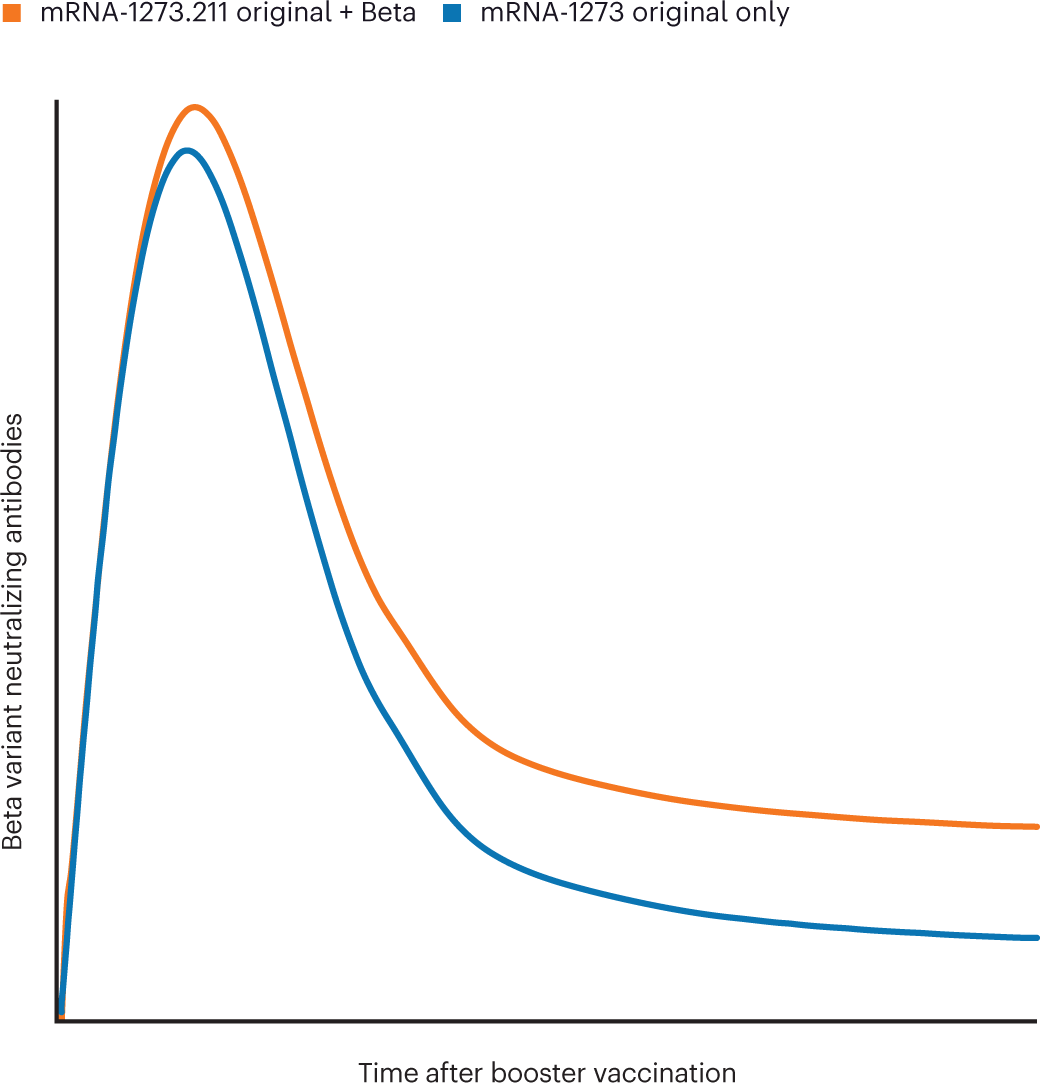

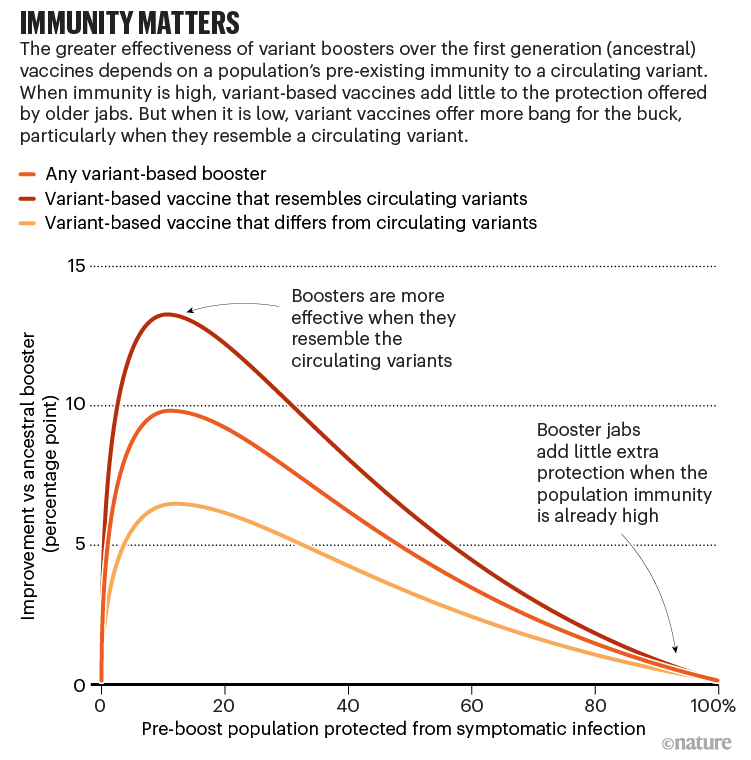
:max_bytes(150000):strip_icc()/Booster-Shot-GettyImages-1362497127-e10ca9b1e3e5424db126022a8dc254bf.jpg)